
Similarly, if you and your pal agree to change the match's length, you can do it with ease. While playing in the online modes against your companions, you can adjust the difficulty levels of hitting and pitching according to your desire in different game modes. Once you have found another online user, you can match with them to play. For this, you should select the 'Locate Teamamtes' tab instead of clicking on the 'Invite Friend' one. Now, if you do not have any known friends in MLB The Show 23, you can still choose to play with random people. Enabling Cross-play is mandatory to play against your cross-platform companions. Once your companion is on your list, you can choose your team, and your companion can choose another. When both have a good internet connection, you can choose any game mode and invite your pal. Many player cards with different rating are present in the Show franchise( Source : Play Against Friends in MLB The Show 23 can be done when they are online and providing invitations to them in the desired game mode. How To Play Against Friends In MLB The Show 23? Tell your friend to accept the invitation.Choose your companion and then send him the link.A link for the Friends list will be presented to you.Select the 'Invite Friend' option in the desired game mode.Select a game mode that you and your pal want to play.Check if your companion is online or not.Here are some of the steps you can follow to invite a companion to your game mode: Players can invite their friends when they are online and need to be on the supported console platforms. Sometimes the consoles may adjust to turn them off for the privacy of not allowing you to communicate with cross-platform users. However, the Cross-play option should be switched on by default. If it is turned off, click on it once, and voila! and you can play with companions from other gaming platforms. Beside the Big 23 logo, there is a Cross-play option on the right side. Here, you will see the big 23 images, with Game Challenges, Unlink Account, and Leaderboards at the bottom. Once you click the icon, then you have to scroll and select the My Profile option. After this, see to the top left side of the screen and select the Profile icon. To turn this option on, you have to go open up your game first and load up the Main Menu. Cross-platform companions will still be affected and should wait for the developer's official solution. You can also go to the co-op screen and invite the other players through the console, but this fix is only for the same platform users. Afterward, log back into The Show 23, which might bring back the companion list. This will enable you and your pal to be featured on the friend list.Īnother solution is to restart your PS device and play another video game for about 30 minutes. 
After you are at the screen, ensure that your companion invites you after your request. 
One way to work around this issue is to request to join the game alongside your pal in the 2v2 ranked menu. 
Ranked co-op is one of the modes to play with your companions.( Source : This problem arose after the new patch update. MLB The Show 23 friends list not working can be fixed through various workarounds for console users. However, another possible solution for the PS players is performing the 'Restore License.' Friends List Not Working In MLB The Show 23 One Reddit user stated that they can still play the game with their pals, who are console users. This works well in line with the Madden games, where people could not play with their pals if one was on Series X while the other was on an Xbox One. However, this fix is only for PS users and won't provide solutions for cross-platform playthroughs. This will enable you to get an invitation to the menu itself. After you select your pal, press the little square, having the controller logo in the middle. For instance, if you are on PlayStation 5, you can go to the PS Menu's game base and then click on which pal to invite to your co-op game. One possible fix for this issue is to invite your friends from PlayStation's Quick Menu. Many Reddit users have also complained about the issues and seek to get an official fix. With the new June update, many players have been facing the problem in co-op mode, where they cannot invite their friends to play games with them. The game has officially licensed baseball players to play with.( Source : Many possible fixes are available for the console users. MLB The Show Friends not showing up is present since the new update.
0 Comments
 
AU-MAL-1900001įor more information on Maltofer, please contact Vifor Pharma Medical Information on or phone 1800 202 674.įor all sales and marketing requests please contact Aspen Pharmacare Australia Pty Ltd, 34-36 Chandos St, St Leonards, NSW 2065. Maltofer® is a registered trademark of Vifor (International) Inc used under licence by Aspen Pharmacare Australia Pty Ltd.ĭate of preparation March 2023. Although not as rapid-acting as iron chelate, its. Iron(III)-hydroxide polymaltose complex in iron deficiency anemia / review and meta-analysis. In horticulture it is used for treating iron chlorosis (yellowing of folliage caused by iron deficiency). Independent study funded by Vifor (International) Inc, St Gallen, Switzerland. Efficacy and safety of oral iron(III) polymaltose complex versus ferrous sulfate in pregnant women with iron-deficiency anemia: a multicenter, randomized, controlled study. 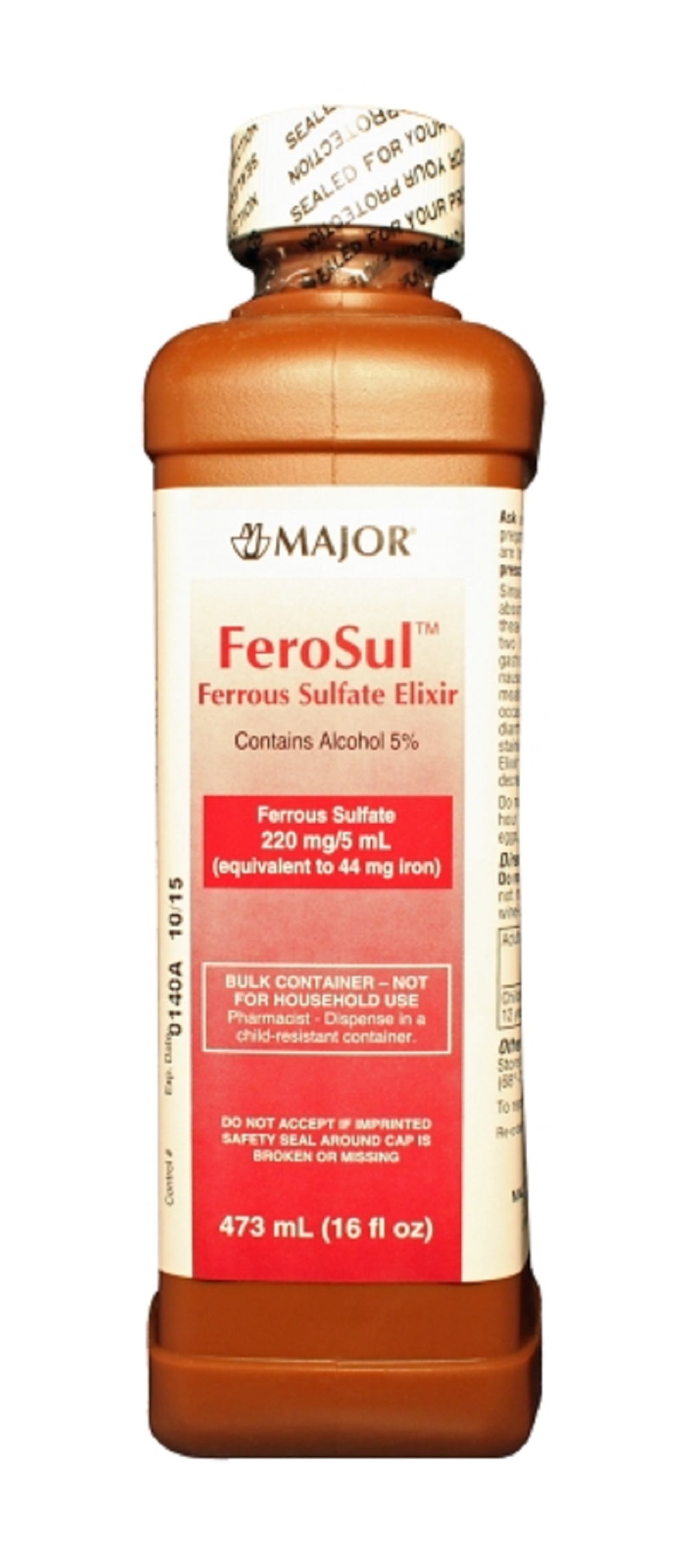
If you have iron deficiency, your doctor will advise you whether an oral iron treatment is required. For the prevention of iron deficiency in adults and adolescents determined by a medical practitioner to be at high-risk, where the use of ferrous iron supplements is not tolerated, or otherwise inappropriate. Ferro-liquid, Ferrous Sulfate 30mg/mL, 250mL Ferro-Liquid is an iron replacement oral liquid solution for iron deficiency and iron deficiency anaemias. Maltofer is for the treatment of iron deficiency in adults and adolescents where the use of ferrous iron supplements is not tolerated, or otherwise inappropriate. ALWAYS READ THE LABEL AND FOLLOW THE DIRECTIONS FOR USE. The loss of electrons results in a positively charged cation, whereas gain forms a negatively charged species called an anion. An ion is formed when an atom loses or gains electrons from its valence electronic orbital to attain a stable electronic configuration. ls = low spin, hs= high spin.Before delving into the definition of ionic radius, it might be necessary to brush up on the basics and discuss the formation of an ion. This is because the greater covalent character of the bonds in AgCl and AgBr reduces the bond length and hence the apparent ionic radius of Ag +, an effect which is not present in the halides of the more electropositive sodium, nor in silver fluoride in which the fluoride ion is relatively unpolarizable. On the basis of the fluorides, one would say that Ag + is larger than Na +, but on the basis of the chlorides and bromides the opposite appears to be true. This is illustrated by the unit cell parameters for sodium and silver halides in the table. No bond is completely ionic, and some supposedly "ionic" compounds, especially of the transition metals, are particularly covalent in character. All compounds crystallize in the NaCl structure.Īn "anomalous" ionic radius in a crystal is often a sign of significant covalent character in the bonding. Unit cell parameters (in pm, equal to two M–X bond lengths) for sodium and silver halides. In general, ionic radius decreases with increasing positive charge and increases with increasing negative charge. Anions (negatively charged) are almost invariable larger than cations (positively charged), although the fluorides of some alkali metals are rare exceptions. 
Ionic size (for the same ion) also increases with increasing coordination number, and an ion in a high-spin state will be larger than the same ion in a low-spin state. As with other types of atomic radius, ionic radii increase on descending a group. Nevertheless, ionic radius values are sufficiently transferable to allow periodic trends to be recognized. The ionic radius is not a fixed property of a given ion, but varies with coordination number, spin state and other parameters. The latter values are thought to be a more accurate approximation to the "true" relative sizes of anions and cations in ionic crystals. Some sources have retained Pauling's reference of r ion(O 2−) = 140 pm, while other sources prefer to list "effective" ionic radii based on r ion(O 2−) = 126 pm. Ī major review of crystallographic data led to the publication of a revised set of ionic radii in 1976, and these are preferred to Pauling's original values. By comparing many different compounds, and with a certain amount of chemical intuition, Pauling decided to assign a radius of 140 pm to the oxide ion O 2−, at which point he was able to calculate the radii of the other ions by subtraction. However, it is not apparent what proportion of this distance is due to the size of the sodium ion and what proportion is due to the size of the chloride ion. 
For example, it can be readily determined that each side of the unit cell of sodium chloride is 564.02 pm in length, and that this length is twice the distance between the centre of a sodium ion and the centre of a chloride ion:Ģ = 564.02 pm X-ray crystallography can readily give the length of the side of the unit cell of a crystal, but it is much more difficult (in most cases impossible, even with more modern techniques) to distinguish a boundary between two ions. The concept of ionic radius was developed independently by Goldschmidt and Pauling in the 1920s to summarize the data being generated by the (then) new technique of X-ray crystallography: it is Pauling's approach which proved to be the more influential. Better weighing performance in 6 easy steps |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
